This biomaterial could help us 3D-print 'while you wait' bones
Rapidly induces bone regeneration
Sign up for breaking news, reviews, opinion, top tech deals, and more.
You are now subscribed
Your newsletter sign-up was successful
Join the club
Get full access to premium articles, exclusive features and a growing list of member rewards.
Every year, thousands of adults and children undergo bone implantation surgery. In adults, metals are sometimes used to replace the bones, but that doesn't work for growing children. So instead bone is sometimes harvested from elsewhere in the body - but that can lead to complications and pain.
What's needed is a synthetic implant that induces natural bone regeneration and growth, which can adapt its shape over time, and whose shape can be easily customised. Now, a research team at Northwestern University, led by Ramille Shah, has developed exactly that.
Biocompatible, Biodegradable
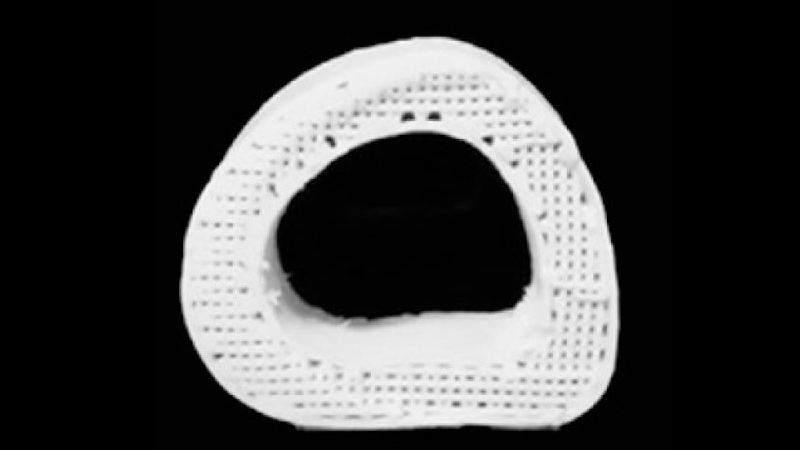
Their solution is a biomaterial that mixes a mineral found naturally in human bone with a biocompatible, biodegradable plastic that's already used in many medical applications. The resulting "bone" material is hyperelastic, strong, and can be printed in a 3D printer at room temperature into whatever shape you need.
Article continues belowMost importantly, it's full of holes. "Porosity is huge when it comes to tissue regeneration, because you want cells and blood vessels to infiltrate the scaffold," Shah said. "Our 3D structure has different levels of porosity that is advantageous for its physical and biological properties."
Further Enhance Regeneration
The benefit of being able to perform the 3D-printing process at room temperature is that other substances can be combined into the ink too. "We can incorporate antibiotics to reduce the possibility of infection after surgery," Shah said.
"We also can combine the ink with different types of growth factors, if needed, to further enhance regeneration. It's really a multi-functional material."
Shah has a vision of a hospital where bones and other implants can be 3D-printed while the patient waits. "The turnaround time for an implant that's specialized for a customer could be within 24 hours," Shah said.
Sign up for breaking news, reviews, opinion, top tech deals, and more.
"That could change the world of craniofacial and orthopaedic surgery, and, I hope, will improve patient outcomes."
The full details of the implant can be found in a paper that the team published in the journal Science: Translational Medicine
- Duncan Geere is TechRadar's science writer. Every day he finds the most interesting science news and explains why you should care. You can read more of his stories here, and you can find him on Twitter under the handle @duncangeere.

 Become a TechRadar Insider
Become a TechRadar Insider






